 Valence electrons are electrons that are present in the valence shell of a molecule, the outermost shell, and the quantity of valence electrons determines an atom's valency (or valence). The general oxidation state of the elements present in the periodic table can be illustrated in the chart provided below. However, these d and f block elements' general valencies are given as 2 and 3. In general, the elements' valencies belonging to both the s-block and the p-block of the periodic table are calculated as eight minus the number of valence electrons or the number of valence electrons.įor the d and f-block elements, valency can be determined not only based on the valence electrons but also on both d and f orbital electrons. At the same time, the number of valence electrons defines the valency or valence of an atom. 
Generally, electrons that are found in the outermost shell are referred to as valence electrons. Oxidation is one of the most fundamental aspects of elements is state and valency, which may be explored using electron configurations. The valency of the element is a measure of its ability to combine with other elements and is defined as the number of electrons that an object must lose or receive in order to achieve a stable electron configuration.The number of electrons lost or acquired by an atom determines its oxidation state. Valency and Oxidation State are the most fundamental properties of the elements and are studied with the electron configurations' help. 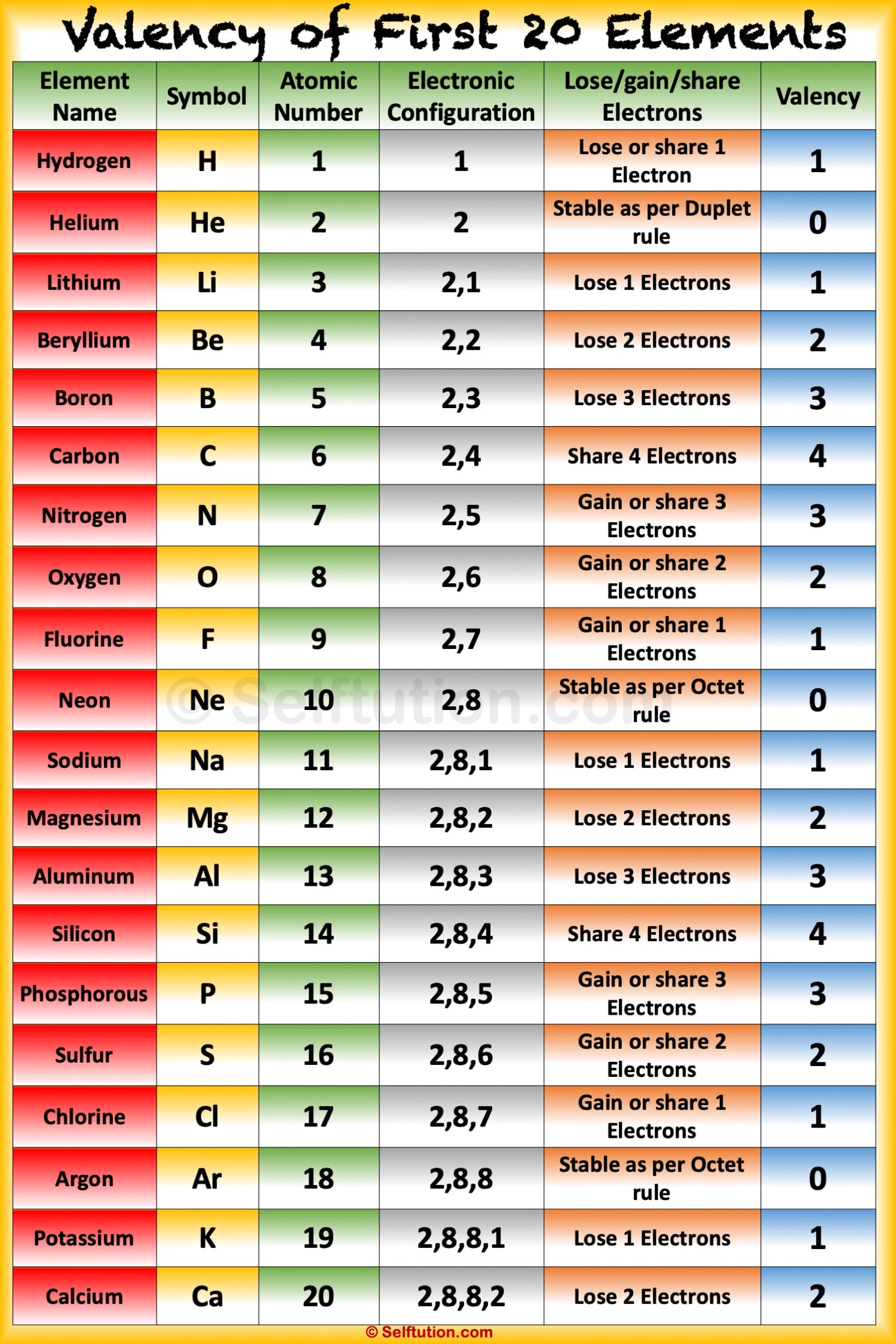
The oxidation state of an atom is defined as the number of electrons gained or lost by it. The valency of an element is described as a measure of its combining capacity, and it can be defined as "the number of electrons that should be gained or lost by an atom to obtain the stable electron configuration." 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
